| References |
| Formal Name |
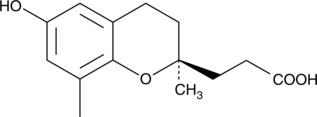
3,4-dihydro-6-hydroxy-2,8-dimethyl-2H-1-benzopyran-2-propanoic acid |
| CAS Number |
1221504-67-1 |
| Molecular Formula |
C14H18O4 |
| Formula Weight |
250.3 |
| Formulation |
A crystalline solid |
| Purity |
>95% |
| λmax |
205, 298 nm |
| Stability |
2 years |
| Storage |
-20°C |
| Shipping |
Wet ice
in continental US; may vary elsewhere
|
| SMILES |
OC(=O)CC[C@]1(C)CCc2cc(O)cc(C)c2O1
|
Background Reading
Kamal-Eldin, A., and Appelqvist, L. The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids 31 671-701 (1996).
Christen, S., Woodall, A.A., Shigenaga, M.K., et al. γ-Tocopherol traps mutagenic electrophiles such as NOx and complements a-tocopherol: Physiological implications. Proc Natl Acad Sci USA 94 3217-3222 (1997).
Chiku, S., Hamamura, K., and Nakamura, T. Novel urinary metabolite of d-δ-tocopherol in rats. J Lipid Res 25 40-48 (1984).
Weiser, H., Riss, G., and Kormann, A.F. Biodiscrimination of the eight α-tocopherol stereoisomers results in preferential accumulation of the four 2R forms in tissues and plasma of rats. J Nutr 126(10) 2539-2549 (1996).
Show all 4
Hide all but first 3
| Size |
Global Purchasing |
| 1 mg |
|
| 5 mg |
|
| 10 mg |
|
| 25 mg |
|
Description
Molecules having vitamin E antioxidant activity include four tocopherols (α, β, γ, δ) and four tocotrienols (α, β, γ, δ).1 One form, α-tocopherol has the highest biological activity based on fetal resorption assays.2 δ-CEHC is a major β-oxidation metabolite of δ-tocopherol.3,4 Approximately 50% of a 3H-δ-tocopherol given as an intraperitoneal dose in rat is recovered in the urine as δ-CEHC, indicating this is the major route of metabolism.4
1
Kamal-Eldin, A., and Appelqvist, L. The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids 31 671-701 (1996).
2
Weiser, H., Riss, G., and Kormann, A.F. Biodiscrimination of the eight α-tocopherol stereoisomers results in preferential accumulation of the four 2R forms in tissues and plasma of rats. J Nutr 126(10) 2539-2549 (1996).
3
Christen, S., Woodall, A.A., Shigenaga, M.K., et al. γ-Tocopherol traps mutagenic electrophiles such as NOx and complements a-tocopherol: Physiological implications. Proc Natl Acad Sci USA 94 3217-3222 (1997).
4
Chiku, S., Hamamura, K., and Nakamura, T. Novel urinary metabolite of d-δ-tocopherol in rats. J Lipid Res 25 40-48 (1984).
|